what phenotypic changes occur to the macrophages that cause them to be anti-inflammatory
- Review
- Open Access
- Published:
Phenotypic alteration of macrophages during osteoarthritis: a systematic review
Arthritis Enquiry & Therapy volume 23, Article number:110 (2021) Cite this article
Abstract
Objective
Osteoarthritis (OA) has long been regarded equally a disease of cartilage degeneration, whereas mounting evidence implies that depression-form inflammation contributes to OA. Among inflammatory cells involved, macrophages play a crucial office and are mediated past the local microenvironment to showroom different phenotypes and polarization states. Therefore, we conducted a systematic review to uncover the phenotypic alterations of macrophages during OA and summarized the potential therapeutic interventions via modulating macrophages.
Methods
A systematic review of multiple databases (PubMed, Spider web of Science, ScienceDirect, Medline) was performed upwards to February 29, 2020. Included manufactures were discussed and evaluated by two independent reviewers. Relevant information was analyzed with a standardized and well-designed template.
Results
A total of 28 studies were included. Results were subcategorized into two sections depending on sources from human tissue/cell-based studies (12 studies) and creature experiments (16 studies). The overall ascertainment indicated that M1 macrophages elevated in both synovium and circulation during OA development, along with lower numbers of M2 macrophages. The detailed alterations of macrophages in both synovium and circulation were listed and analyzed. Furthermore, interventions confronting OA via regulating macrophages in animal models were highlighted.
Decision
This study emphasized the importance of the phenotypic alterations of macrophages in OA evolution. The classical phenotypic subcategory of M1 and M2 macrophages was questionable due to controversial and conflicting results. Therefore, further efforts are needed to categorize macrophages in an exhaustive manner and to utilize advanced technologies to identify the private roles of each subtype of macrophages in OA.
Introduction
Osteoarthritis (OA) is the nearly common degenerative articulation disorder, mainly affecting the weight-begetting joints such as knees and hips [1], and the non-weight-begetting joints such as the hand and temporomandibular joints [ii]. OA is the main crusade of physical inability and has been predicted to afflict approximately 67 million people in U.s.a. by 2030 [iii]. Though gamble factors such as aging, obesity, genetic predisposition, and articulation trauma have been identified for OA initiation [4,5,six,7], few effective treatments are bachelor to prevent OA due to the insufficient agreement of the pathogenesis [eight]. Recently, accumulating evidence indicates that the inflammation significantly contributes to OA in addition to the abnormal mechanical loading [9]. OA is gradually viewed as a low-grade inflammatory illness affecting the whole joint likewise articular cartilage [ten]. Collectively, these findings highlight the profound function of innate immunity in the progression of OA.
As a disquisitional office of the innate allowed organisation, macrophage has long been considered as an important participant in OA. For example, depletion of synovium macrophages by magnetic chaplet (anti-CD14-conjugated magnetic beads) [11] or chemicals (eastward.g., clodronates) [12] contributes to decreasing cartilage catabolic enzymes such as MMP13 and Adamts4. Withal, the systemic ablation of macrophage in MaFIA (Macrophage Fas-Induced Apoptosis) mice leads to a severe synovitis in obese OA model, implying the complicated roles of macrophages in OA [13].
Macrophages play diverse roles in development, inflammation, and tissue repairing. The plasticity of macrophages enables the cells to brand adjustments towards local microenvironments and respond to a wide range of stimuli under both physiological and pathological weather condition [14, 15]. Studies in ferreting out the role of macrophages in inflammation have progressed recently. Researchers take identified 2 different polarization status of macrophages when confronted with different stimuli. In inflammatory phase, classically activated M1 macrophages are recruited and produce high levels of pro-inflammatory cytokines and chemokines, such as tumor necrosis gene-alpha (TNF-α), IL-1, and IL-half-dozen. On the other hand, alternatively activated M2 macrophages are needed for the resolution of inflammation. M2 macrophages reverse the inflammatory procedure by releasing anti-inflammatory factors such as IL-ten and secreting growth factors, such as transforming growth factor-beta (TGF-β) [16]. Therefore, polarization of macrophages at different inflammatory stages might account for various pathological processes. Although the concept of macrophage M1/M2 polarization provides an effective system to study macrophages in vitro, the exact definition and phenotypic transition of macrophages in in vivo studies are nonetheless less divers [17].
As mentioned, emerging studies aimed to control inflammation in different diseases by targeting phenotypic changes of macrophages. In pre-clinical models, normalizing the aberrant M1/M2 ratio has been suggested as a therapeutic strategy for macrophage-involved diseases, such as atherosclerosis, lung cancer, and os diseases including osteoporosis and osteoarthritis [18,19,20,21,22].
In this study, we systematically reviewed contempo key findings of macrophage polarization in OA, evaluated the role of phenotypic alterations in macrophages, and summarized the current and potential interventions via the modulations of macrophages.
Methods
Search strategy
A systematic search was set on PubMed, WOS (Web of Science), Ovid (MEDLINE database), Embase (Elsevier Database), and Scientific discipline Direct (Elsevier Databases), according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [23]. It was performed without whatever limitation to the publication date in guild to identify all manufactures on the role of M1 and M2 macrophages in OA. The medical term for osteoarthritis was used in combination with phenotypic alterations of macrophages, which was shown below: osteoarthritis [Title/Abstract] AND macrophage [Title/Abstruse] AND (Polarization [Title/Abstract] OR polarization [Title/Abstract] OR M1 [Title/Abstruse] OR M2 [Title/Abstract] OR inflammation [Title/Abstruse]). The search was last updated on February 29, 2020.
Screening process
All articles were screened by ii contained investigators (XBZ and HTX). The reviewing option procedure was based on title, abstract, and full-text level, using a well-established screening tool, Covidence [24].
The eligibility of each article was adamant co-ordinate to the inclusion and exclusion criteria. Studies were included if they met the post-obit criteria: (i) relevant to the searching strategy, (ii) English language-written articles within the recent 20 years, and (ii) availability of full-length inquiry manufactures. Exclusion criteria were nowadays as following: non-English written studies, case studies, review articles, editorials, messages, conference paper, or book chapters. Moreover, the contents of the manufactures were also taken into consideration. Studies that were not related to OA or without measurements of macrophages were excluded.
Studies meeting all criteria were included, and the quality cess of these articles was based on heterogeneity and methodological quality. Methodological qualities of included studies were based on the quality systems, which were raised by Wells et al. [25,22,23,28]. Details almost the qualities of the included studies were listed in Supplemental materials.
At each step of screening, a final consensus was reached later on the mutual discussion between ii investigators (XBZ and HTX) when dissimilar opinions existed. Information were extracted and tabulated by XBZ, and then, a subset of key variables was validated by HTX.
All included articles were double-checked by the third investigator (YFW). Similarly, relevant data were extracted and analyzed with a standardized method designed for this review (see Fig. 1. for the flowchart).
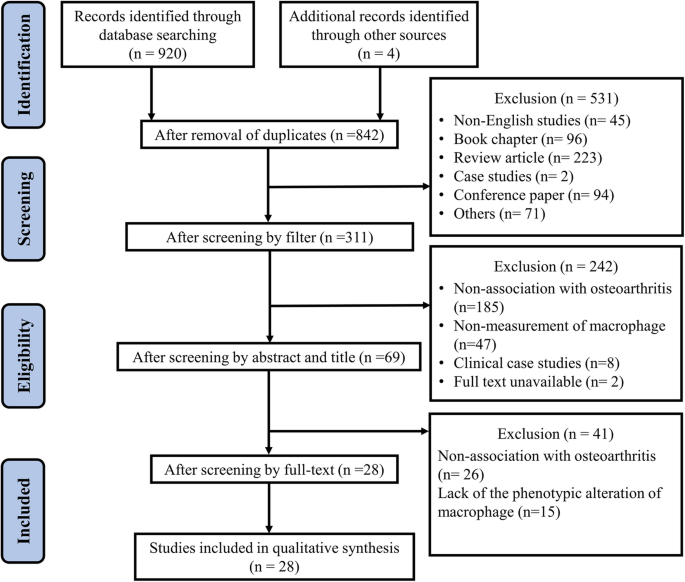
Flowchart of studies included in the systematic review. After the application of all inclusion and exclusion criteria, 28 studies were identified for analysis
Results
Of the 920 manufactures screened that were considered to exist relevant to the inquiry topic, 28 manufactures were finally included. The screening process is available in Fig. 1. The detailed information of the 28 selected manufactures are shown in Tabular array 1. In this review, the included articles were categorized into two sections based on the species every bit different species have dissimilar cellular compartments [56]. The distributional proportions of all species in included articles are shown in Fig. 2a. Amongst all included articles, studies focusing on tissues or cells from homo species constituted less than half of total (12/28), followed by animate being studies (xvi/28), including mouse (ix/28), rat (4/28) and other animals (canine and equine studies, three/28). Inside these two subcategories (homo-derived and animal studies), the origin of macrophages was besides evaluated, particularly for the peripheral macrophages (peripheral blood monocyte-derived macrophages) and the tissue-resident macrophages (synovial macrophages). In each subcategory, further evaluation was conducted on the interventions that could touch on or modulate the M1/M2 ratio in OA progression.

Feature outcomes of experimental species, animal models, and species distribution in beast interventional studies. a The proportion of experimental species. 43% studies (12/28) were based on chief cells or samples from human. 32% studies (nine/28) were based on chief cells or samples from mice. 14% studies (4/28) were based on main cells or samples from rats. 11% of studies (three/28) were based on samples from canine and equinel; b Pie chart illustrating the types of animal models used in the studies. 36% the fauna models were ACLT (4/11). 36% the animal models were DMM (4/11). eighteen% the creature models were intra-articular injection of collagenase (2/11). 9% the animal models were other beast models including osteochondral defect model (1/11); c Species distribution in interventional animal studies. 64% the interventional studies were based on mouse models. 36% the interventional studies were based on rat models
Man tissue/prison cell-based studies
In mammals, most organs contain tissue-resident macrophages. These macrophages are believed to be distributed into unlike tissues during embryogenesis [15] and are essential for maintaining allowed homeostasis. On the other hand, circulating monocyte-derived macrophages also play a crucial office in inflammation. In response to inflammation, circulating monocytes are recruited to inflamed tissues and afterwards differentiate into macrophages in situ past local inflammatory mediators [57]. For healthy individuals, a small number of macrophages constitutively reside in the human knee joint. Still, in the OA knees, the intensity and infiltrated areas of macrophages in synovium are significantly increased [58].
M1 and M2 macrophage in the peripheral blood from OA patients
Studies of peripheral blood monocytes (PBMCs) in OA were carried out in the by few years. PBMCs would partially polarize towards M1 or M2 macrophages after stimuli, and the phenotypic alterations of macrophages were identified based on cellular markers.
In i study, PBMCs were isolated and induced to differentiate into macrophages, and and so, CD14+CD11b+ macrophages (M0, naive macrophages) were purified and selected. Upon stimulated with OA-related metabolite, such as basic calcium phosphate (BCP) crystals, macrophages produced college levels of chemokines, such as CXCL9 and CXCL10, and increased the expression of M1 surface markers, such as CD86 and CD40 [29]. These results unsaid that the PBMCs tended towards the M1 phenotype during OA.
In some other homo prison cell-based written report, the proportion of pro-inflammatory CD11c+ macrophages was significantly higher in the circulation of OA patients than that of healthy individuals. In dissimilarity, the number of anti-inflammatory CD206+ macrophages was markedly lower in OA patients [34]. These results revealed a higher M1/M2 ratio in the peripheral blood of OA patients.
M1 and M2 macrophage in synovium from OA patients
Synovium-resident macrophages were activated in OA and contributed to the principal source of cytokines in OA progression [59].
Researchers accept identified the abnormal accumulation and phenotypic alterations of macrophages in OA synovium [44, 49], coordinating to those in the peripheral blood. Compared to salubrious synovium, OA synovium demonstrated a marked elevation of F4/eighty+ (macrophage marking) cells in both intimal and sublining layers, together with a higher number of iNOS+ (M1-like macrophage marking) cells in the intimal lining layer. Conversely, CD206+ (M2-like macrophage marker) cells showed only a slight simply non-meaning subtract in OA synovium [37].
Hoffa's fatty pad (HFP) is another macrophage niche in the articulatio genus joints [60]. Timur et al. divided the OA conditions into two groups according to the level of Prostaglandin E2 (PGEii) in HFP explant civilisation medium (100 mg fat pad tissue /ml): high PGE2 grouping (> 25 ng/ml) and low PGEtwo group (< 25 ng/ml). The HFP from high PGE2 OA group showed a 21.iv-fold college inflammation ratio than HFP from low PGEtwo OA group. Meanwhile, HFP from high PGE2 OA grouping demonstrated 3.vii-fold lower cistron expression of CD163 (M2 macrophage marking) compared to the HFP in low PGE2 group. These results indicated that the role of macrophage polarization might vary in different OA subtype [38].
In vitro human tissue/cell-based study
Although the in vitro data from isolated cells or tissues are not yet ready to be translated into clinical interventions in OA. Several in vitro studies with exogenous treatment still showed potentials to normalize the macrophage polarization and protect against OA. For example, overexpression of CD163 in primary man macrophages by a polyethylenimine nanoparticles grafted with a mannose ligand (Human being-PEI) contributed to the transition of macrophages from M1 to M2 later beingness stimulated with LPS [51]. Pal et al. validated that sulforaphane (10 μM) could skew the differentiation of monocytes (man monocyte prison cell line: THP1 cells; human primary monocytes) towards the anti-inflammatory M2 type [41]. Some other sample is lumican (LUM), a major extracellular matrix glycoprotein in articular cartilage, and its expression was significantly upregulated in OA [48]. LUM contributed to the innate immune-mediated pathogenesis of primary OA via promoting macrophage M1 polarization [48], and this made LUM get a promising therapeutic target for OA. In improver, the human adipose-derived MSCs were reported to be responsible for phenotypic switching from M1 to M2 in human macrophages, accompanied with the decreased secretion of inflammatory cytokines such as TNF-α and IL-vi [40]. These cellular and molecular mechanisms are related to the modification of macrophages in OA, and the treatments have shown therapeutic effects in human in vitro studies.
Animate being studies
Due to the complication of OA, at that place are still many unknowns in pathogenesis of OA. For example, the exact trigger for initiating the cartilage degradation is still unknown. As well, the underlying mechanisms that atomic number 82 to disease maintenance rather than resolution are yet poorly understood. Therefore, animal studies go valuable to delineate the underlying mechanisms of the disease and develop novel therapies.
In this section, we reviewed the articles on the phenotypic alterations of macrophages in dissimilar species and different animal models. Since there are many dissimilar types of OA animal models, the proportion of unlike OA models applied in the included studies is shown in Fig. 2b.
M1 and M2 macrophages in synovium from experimental animals
Destabilization of medial meniscus (DMM)-induced OA, a well-established OA model [61], has been widely used to written report the macrophage alterations in OA. Four studies were included with DMM model in this department, and all studies reported a significantly higher number of synovial F4/fourscore+ cells mail DMM, indicating that the innate immune system was activated during DMM-incurred OA progression. One study drew the conclusion that the number of M1 macrophages (F4/80+CD86+CD63−) was significantly increased in murine synovium half dozen weeks afterwards DMM [36]. In another written report, synovial F4/lxxx+CD11c+CD206+ cells significantly decreased later on DMM for both 4 and eight weeks [35]. Additionally, another study showed a tendency towards more NOS2+ cells (M1 macrophages) in the DMM-operated joint than those in contralateral articulation [36]. Like trends in the change of macrophage populations were also found in a more than astringent OA model, inductive cruciate ligament transection (ACLT)-induced rodent OA model [30, 47].
Furthermore, there were three studies in large animals. I was a canine OA model [32], and ii were equine OA models [53, 62]. In the canine study, synovial fluid samples were collected and analyzed, and researchers discovered that ratio of positively stained M1-polarized macrophages (CCR7+iNOS+CD68+ cells) to M2-polarized macrophages (CD163+Arg1+CD68+ cells) was higher in OA group than that in the normal control group [32]. In an ex vivo study, the equine osteochondral-synovial explant co-culture system was facilitated as an OA model. Researchers evaluated the ratio of NO (μM)/urea (μM) as a symbol of pro-inflammatory M1-like macrophages as previously described [63] and concluded that the macrophages underwent a shift towards M1 phenotype during OA progression [33]. Recently, another study demonstrated that os marrow-derived mononuclear cells, which were viewed equally a source of macrophages, displayed an M2-like transcriptional characteristics when stimulating with inflamed synovial fluid in an equine OA synovitis model (0.five ng LPS injection into radiocarpal joint for viii h) [53].
Information technology was too noteworthy that though the phenotypic alterations of macrophages during rheumatoid arthritis (RA) had already been discovered recently [64, 65], in that location was however a knowledge gap in the phenotypic alterations of peripheral claret-derived monocytes or macrophages in OA animal models.
Interventions in animal models
Amidst all included animal studies, xi studies were selected and highlighted for the interventions against abnormal changes of macrophages. As shown in Fig. 2c, 64% of these studies (7/11) were based on the mouse models while 36% of the studies (4/xi) were based on the rat model. Researchers utilized diverse methodologies to correct the aberrant macrophage polarization (Fig. 3 and Table 2), including interventions regulating and targeting specific signaling pathways [31, 37, 52, 54] and comprehensive interventions such as extracts from traditional Chinese herbs [30, 46, 47], anti-inflammation drugs [42], mesenchymal stem cell (MSC)-related therapies [45, 55], and others [36].
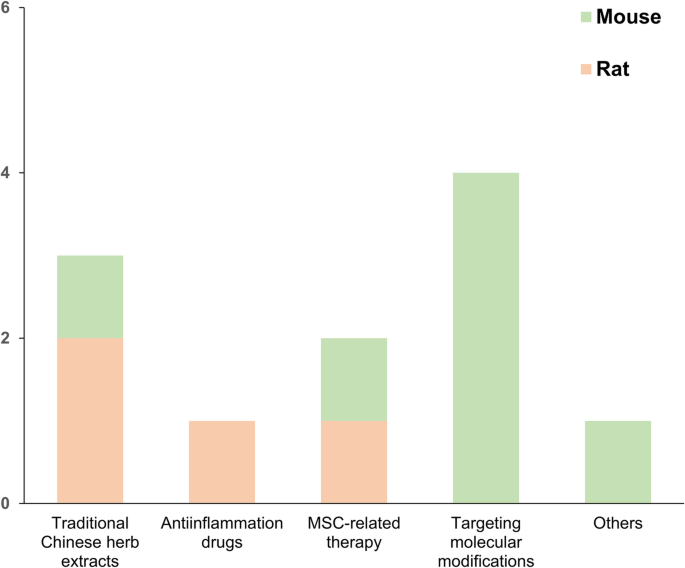
Interventional subcategories. The interventions against OA included traditional Chinese herb extracts, anti-inflammation drugs, MSC-related therapy, targeting molecular modifications, and others
For animal OA models, various traditional Chinese herb extracts such as kinsenoside, quercetin, and squid type II collagen were identified to exist able to repolarize the synovial macrophages and attenuate cartilage degeneration in OA [thirty, 46, 47].
Moreover, to further elucidate the endogenous immunological mechanism of OA, endogenous molecules and the relevant signaling pathways were identified. Mammalian target of rapamycin circuitous ane (mTORC1) signaling pathway was reported to be aberrantly activated in synovial macrophages during OA and afterwards contributed to macrophage M1 polarization. These polarized macrophages (M1) produced excessive R-spondin-2 (Rspo2) and and then exacerbated experimental OA [37]. By intra-articular injection of Rspo2 neutralizing antibody, the cartilage degeneration incurred past M1 macrophage polarization was effectively ameliorated [37]. In addition, activating silent information regulator 2 ortholog 1 (Sirt1) signaling pathway with a selective Sirt1 activator SRT210 relieved DMM-induced OA, partially through normalizing the synovial macrophage polarization (increased the CD206+ M2 macrophages and decreased the iNOS+ M1 macrophages) [52]. With the evolution of nanotechnology, a modified nanoparticle (NP) termed zeolitic imidazolate framework-8 nanoparticle (ZIF-8 NP) was designed to target synovial macrophages and transform macrophage polarization from M1 to M2 phenotype, thus attenuating OA [54]. These interventions and signaling pathways are integrated in Table 3.
Stem prison cell-based therapies were also found to salve OA via modulating macrophage activation [66]. The awarding of stem cells for cartilage repair relied on their ability to differentiate into chondrocytes and and then substitute for the degenerative or expressionless chondrocytes [67]. Recent studies revealed that the reparative potential of MSCs for OA was too based on its immunological modification on the macrophages [39, 68]. The assistants of man embryonic stem cell-derived exosomes in the osteochondral defect model could increase the CD163+ macrophages (M2) and subtract the CD86+ macrophages (M1) in the joint, and lowered the levels of inflammatory cytokines inside the joint (cartilage and synovium) [68, 69]. Inversely, as a feedback, the inflammatory microenvironment also impacted chondrogenic differentiation of mesenchymal stalk cells via macrophage polarization [43].
Discussion
Recently, the crucial role of synovium resident macrophages in the pathogenesis of RA has been well recognized. Depletion of macrophages or normalizing macrophage phenotype could protect against RA [seventy,67,72], and the exact role of M1 and M2 macrophages in RA has also been well reviewed [73]. However, footling has been systematically reviewed on the part of macrophages and their polarization in OA. In this study, we systematically reviewed the properties of M1 and M2 macrophages in the periphery, synovial tissue, and synovial fluids of OA patients. We presented important findings from both human and animal OA models and summarized the relevant targeting interventions or comprehensive interventions in these studies, which gave rise to normalizing the phenotype of macrophage (M1 or M2) and alleviating the OA progression.
All the same, some experimental results were relatively contradictory, which suggest that macrophages take multifaceted and complicated roles in OA. In a DMM-induced OA model in obese mice (high-fatty nutrition for x weeks and 20 weeks), Wu et al. eliminated macrophages globally with a pocket-sized molecule named AP20187 in MaFIA mice. The macrophage-deleted mice immediately exhibited less osteophyte formation following DMM, merely these obese mice failed to relieve cartilage deterioration at week 9 and synovial inflammation was also activated in macrophage-depleted mice compared to that in not-depletion mice at week 9 [13]. These observations were inconsistent with previous studies conducted by Blom et al. that deletion of macrophages via intra-articular injection of clodronate liposomes could relieve the inflammatory response and cartilage catabolic enzymes in a collagenase-induced OA model in C57Bl/half-dozen mice [12, 74]. Further, Wu et al. also found that CD3+ T cells and neutrophils massively infiltrated into the DMM-operated articulatio genus and caused severe joint synovitis [thirteen]. The above study emphasized the potential roles of macrophage in maintaining joint homeostasis after injury apart from pro-inflammation and implied that macrophage would participate in limiting the adaptive allowed response that developed after the initial innate response. Thus, it was capricious to treat OA past merely deleting whole macrophages in the joint, without discriminating the dissimilar subtypes of macrophages. Further attention should exist paid to identifying and mobilizing the potential positive roles of macrophages on cartilage repair in OA.
Coincidently, Zhang et al. furthered the research on identifying the roles of different phenotypes of macrophages in OA [37]. They demonstrated that M1 but not M2 macrophages accumulated in synovial tissues from human OA and murine collagenase-induced OA. Activating synovial macrophage M2 polarization by ablating an upstream activator of mTORC1 pathway (i.eastward., Ras homolog enriched in brain 1/Rheb1) in myeloid lineage cells prevented OA evolution. Conversely, deleting an upstream inhibitor of mTORC1 pathway (i.due east., tuberous sclerosis complex i/TSC1) in myeloid lineage cells enhanced M1 polarization and exacerbated cartilage harm in both surgically induced OA and collagenase-induced OA [37]. Furthermore, they identified R-spondin-2 (Rspo2) equally an M1 macrophage-produced protein involved in the subsequent OA progression. Thus, clinical attention could be paid to the mTORC1 signaling pathways in synovial macrophages, and the assistants of Rspo2 inhibitor or neutralizing antibiotic in the articulatio genus joints. Based on Zhang's study, the previous conflicting results (Blom'southward and Wu'south studies) that global depletion of macrophages failed to prevent OA might be partially explained. We made the speculation that information technology was the phenotypic shift (e.thou., from M0 to M1 by downregulating TSC1; from M0 to M2 by downregulating Rheb1), other than the number of macrophages that accounted for the alien results.
Understanding phenotypic changes of macrophage polarization and the function of each subtype would shed low-cal on the potential clinical implication of the interventions. For example, regenerative therapies such equally human embryonic stalk prison cell-derived exosomes showed potential to preclude cartilage deterioration via activating the pro-survival Akt signaling pathway, and reprogramming articulation macrophage by increasing M2 transition and decreasing M1 macrophage infiltration [45]. Intriguingly, a recently adult nanoparticle (modified ZIF-viii NP) showed the similar therapeutic effects on cartilage protection and macrophage repolarization (described in 3.2.2) [54]. Thus, although the macrophage repolarization in OA is still under pre-clinical investigation, it is of great value to attach more than importance to regulating macrophage polarization with the respect to the potential clinical awarding and successful awarding cases in other skeletal illness such as RA [72, 75].
Emerging improvements regarding the correlation of OA and macrophages have been fabricated these years, just a number of unanswered questions were remained. Firstly, the classical M1/M2 classification was bereft for researchers to describe and explicate the fragile mechanisms, and the subtypes of macrophages were required to be delineated strictly. Although several subtypes of M2 macrophages (e.thousand., M2a, M2b, M2c, M2d) [76] were categorized based on the in vitro inducing cytokine combinations (i.e., M2a: IL-iv and IL-13; M2b: LPS and allowed complex; M2c: IL-10 and glucocorticoids; M2d: IL-6) [77,74,79]. Researchers still unintentionally overlooked the sub-classification of macrophages in vivo. Only one written report focused on the M2c macrophage in OA and concluded that hyaluronan could significantly increase the number of anti-fibrotic M2c macrophages (F4/fourscore+CD206+CD301+) 12 weeks post DMM [55]. Farther investigations are thus needed.
The materials and methods in included studies covered the most commonly used molecular biological technologies (shown in Table 2). The phenotypic alterations of macrophage in OA were effectively identified with these technologies, both transcriptionally and translationally. However, the detective methodologies and cellular markers of M0, M1, and M2 macrophages varied and had limitations, and impacted the data interpretation among studies and research models. For instance, the F4/fourscore was widely adopted equally a cellular marking for macrophage in many included studies, but the specify and sensitivity is not that satisfying [80]. The expression of F4/fourscore varied amongst mouse mononuclear populations, being very low in the bone marrow Ly6c+ monocyte-emanated macrophage, but high in diverse tissue-resident macrophage derived from fetal yolk sac, such every bit microglia (brain) and Kuffer cells (liver) [80]. We summarized the cellular markers employed in the included studies in Table 4. In the future, the booming evolution of unmarried-cell methodologies volition help to place the individual cell types at a single-cell level, which could exist used to target distinct macrophage populations precisely.
Although standardization of M1/M2 phenotype gave researchers a uniform framework to written report macrophages in vitro and made results from unlike studies comparable, several drawbacks were all the same concerned. The rigid subdivision of macrophage equally M1 and M2 hindered the understanding of macrophage plasticity in vivo, since classical M1 and M2 polarization were 2 distinct macrophage subtypes and they were unlikely to occur in a tissue context. For instance, the classical M1/M2 image failed to explicate the different transcriptomic changes of man monocyte-derived macrophages and lung-resident alveolar macrophages after stimulated with LPS/IFN-γ (M1 inducer) or IL-4/IL-xiii (M2 inducer) [81]. These macrophage responses indicated that there remained room for macrophages to be further grouped. Avraham et al. conducted a single-jail cell RNA-sequencing to testify that peritoneal macrophages in the same microenvironment respond differently to the stimulus of salmonella strains that differed by a single factor termed PhoP [82].
The classical concept of the M1/M2 paradigm might exist misleading, especially at early stage of inflammation, when tissue-resident and monocyte-derived macrophages coexisted in the local microenvironment. Tissue-resident macrophages expressed relatively higher levels of "M2-similar" markers in comparison with mature monocyte-derived macrophages [83, 84]. Without lineage tracing markers, the simultaneous presence of macrophage population in early inflammation (immature monocyte-derived macrophages and few mature tissue-resident macrophages) would be regarded as an "M1-like" polarization trend due to more young monocyte-derived macrophages in the mixed population. During weeks-long resolution, monocyte-derived macrophages gradually matured and their phenotype resembled the tissue-resident macrophages. Therefore, the mixed cellular population (mature monocyte-derived and mature tissue-resident macrophages) would display a "shift" towards an "M2-like" phenotype [84]. Thus, in the in vitro study conducted by Menarim et al. [53], nosotros speculated that stimuli (SF or ISF) might fail to mobilize the monocyte-derived macrophages from circulation and incur the insufficiency of the M1-like macrophage.
Recently, Culemann et al. plant that in healthy murine and human knee joint, a thin layer of synovium-resident macrophage formed a barrier-like construction, covering the sublining layer of synovium. Disrupting this construction by genetic depletion or pharmacological inhibition of these bulwark-forming macrophages exacerbated arthritis progression. Farther tracing of these macrophages revealed that these CX3CR1+ lining macrophages were originated from a subtype of synovium-resident interstitial macrophage, instead of monocytes [65]. Although these results were discovered in RA model, information technology also unsaid that the critical roles of the synovium-resident macrophage or subchondral bone-resident macrophage is worthy of investigation in OA. They were also the potential cellular targets for further therapeutic design. The recently emerging applications of single-cell RNA sequence or mass cytometry could dissect the exact macrophage phenotypes more than M1/M2 paradigm [57, 85].
Limitation
The quality of included studies was limited due to the low number of high-level evidence. Every bit mentioned in the discussion, the majority of included studies exerted classical methodologies, thus lacking deep insights into the phenotypic alterations of dissimilar subtypes of macrophages.
Secondly, the spontaneous OA models were non included due to lack of reports. For example, crumbling model in Guinea pig recapitulates the nigh mutual cause of man OA [86]. Researches using this model will heighten the translational value in future study.
Thirdly, due to lack of standardized and well-recognized marker for identifying different subtypes of macrophages, the results of different studies were non very comparable.
Lastly, the sample size included in our study was modest, both in the number of articles included and the number of animal samples used in some included studies. The small sample size made it at the risk of bias and difficult to reach general conclusions.
Decision
In summary, we reviewed the electric current studies of phenotypic alterations of macrophages in OA and emphasized the disequilibrium of M1 and M2 macrophage during OA, and potential therapies that could rebalance between M1/M2 macrophages. With a more thorough agreement of macrophages and the improvement in detecting methodology, the detailed macrophage subtypes and their individual roles in OA pathogenesis should be farther elucidated.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- ACLT:
-
Anterior cruciate ligament transection
- BCP:
-
Basic calcium phosphate
- BMMC:
-
Bone marrow mononuclear cells
- DMM:
-
Destabilization of medial meniscus
- HFP:
-
Hoffa's fatty pad
- IL-i:
-
Interleukin-1
- IL-six:
-
Interleukin-6
- Lum:
-
Lumican
- MaFIA:
-
Macrophage Fas-Induced Apoptosis
- mTORC1:
-
Mammalian target of rapamycin complex 1
- NP:
-
Nanoparticle
- OA:
-
Osteoarthritis
- PBMC:
-
Peripheral blood monocytes
- PRISMA:
-
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- RA:
-
Rheumatoid arthritis
- Rheb1:
-
Ras homolog enriched in brain 1
- Rspo2:
-
R-spondin-2
- PGE2 :
-
Prostaglandin E2
- Sirt1:
-
Silent data regulator 2 ortholog 1
- SMM:
-
Synovium-derived macrophage
- TGF-β:
-
Transforming growth factor-beta
- TNF-α:
-
Tumor necrosis gene-blastoff
- TSC1:
-
Tuberous sclerosis complex one
- WOS:
-
Web of Science
References
-
Chu CR, Millis MB, Olson SA. Osteoarthritis: from palliation to prevention: AOA critical issues. J Bone Joint Surg. 2014;96:e130. https://doi.org/10.2106/JBJS.Thousand.01209.
-
Hunter DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet. 2019;393(10182):1745–59. https://doi.org/x.1016/S0140-6736(19)30417-9.
-
Hootman JM, Helmick CG. Projections of Us prevalence of arthritis and associated activity limitations. Arthritis Rheum. 2006;54(1):226–9. https://doi.org/10.1002/art.21562.
-
Mobasheri A, Rayman MP, Gualillo O, Sellam J, Van Der Kraan P, Fearon U. The role of metabolism in the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2017;13(5):302–11. https://doi.org/x.1038/nrrheum.2017.50.
-
Chen D, Shen J, Zhao West, Wang T, Han Fifty, Hamilton JL, et al. Osteoarthritis: toward a comprehensive understanding of pathological mechanism. Os Res. 2017;v:16044. https://doi.org/10.1038/boneres.2016.44.
-
Zhu X, Chen F, Lu M, Wei A, Jiang Q, Cao W. PPARγpreservation via promoter demethylation alleviates osteoarthritis in mice. Ann Rheum Dis. 2019;78(10):1420–9. https://doi.org/ten.1136/annrheumdis-2018-214940.
-
Loeser RF, Collins JA, Diekman BO. Ageing and the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2016;12:412–20. https://doi.org/10.1038/nrrheum.2016.65.
-
Oo WM, Hunter DJ. Affliction modification in osteoarthritis: are we there notwithstanding? Clin Exp Rheumatol. 2019;37:135–xl.
-
Orlowsky EW, Kraus VB. The function of innate immunity in osteoarthritis: when our first line of defence force goes on the offensive. J Rheumatol. 2015;42:363–71. https://doi.org/x.3899/jrheum.140382.
-
Robinson WH, Lepus CM, Wang Q, Raghu H, Mao R, Lindstrom TM, et al. Low-grade inflammation as a key mediator of the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2016;12:580–92. https://doi.org/x.1038/nrrheum.2016.136.
-
Bondeson J, Wainwright SD, Lauder S, Amos N, Hughes CE. The role of synovial macrophages and macrophage-produced cytokines in driving aggrecanases, matrix metalloproteinases, and other destructive and inflammatory responses in osteoarthritis. Arthritis Res Ther. 2006;eight:1–12. https://doi.org/10.1186/ar2099.
-
Blom AB, Van Lent PL, Libregts South, Holthuysen AE, Van Der Kraan PM, Van Rooijen N, et al. Crucial part of macrophages in matrix metalloproteinase-mediated cartilage destruction during experimental osteoarthritis: involvement of matrix metalloproteinase iii. Arthritis Rheum. 2007;56:147–57. https://doi.org/10.1002/art.22337.
-
Wu C-L, McNeill J, Goon K, Trivial D, Kimmerling K, Huebner J, et al. Conditional macrophage depletion increases inflammation and does not inhibit the development of osteoarthritis in obese macrophage Fas-induced apoptosis-transgenic mice. Arthritis Rheumatol. 2017;69:1772–83. https://doi.org/10.1002/art.40161.
-
Li Grand, Nandakumar KS. Macrophages in the activation and resolution of inflammation. In: Nandakumar KS, editor. Autoimmune disorders: risk factors, pathogenesis and treatments, vol. Chapter 6. U.s.a.: Nova publishers; 2019. p. 215–43.
-
Davies LC, Jenkins SJ, Allen JE, Taylor PR. Tissue-resident macrophages. Nat Immunol. 2013;14:986–95. https://doi.org/ten.1038/ni.2705.
-
Huynh M-LN, Fadok VA, Henson PM. Phosphatidylserine-dependent ingestion of apoptotic cells promotes TGF-β1 secretion and the resolution of inflammation. J Clin Invest. 2002;109:41–l. https://doi.org/10.1172/JCI11638.
-
Martinez FO, Gordon Southward. The M1 and M2 paradigm of macrophage activation: fourth dimension for reassessment. F1000Prime Rep. 2014;half-dozen:13. https://doi.org/x.12703/P6-xiii.
-
Bailey KN, Furman BD, Zeitlin J, Kimmerling KA, Wu C-L, Guilak F, et al. Intra-articular depletion of macrophages increases acute synovitis and alters macrophage polarity in the injured mouse knee joint. Osteoarthr Cartil. 2020;28(5):626–38. https://doi.org/10.1016/j.joca.2020.01.015.
-
Momtazi-Borojeni AA, Abdollahi Eastward, Nikfar B, Chaichian Southward, Ekhlasi-Hundrieser M. Curcumin as a potential modulator of M1 and M2 macrophages: new insights in atherosclerosis therapy. Heart Fail Rev. 2019;24:399–409. https://doi.org/x.1007/s10741-018-09764-z.
-
Yuan A, Hsiao Y-J, Chen H-Y, Chen H-Westward, Ho C-C, Chen Y-Y, et al. Opposite effects of M1 and M2 macrophage subtypes on lung cancer progression. Sci Rep. 2015;5:14273. https://doi.org/ten.1038/srep14273.
-
Dou C, Ding North, Zhao C, Hou T, Kang F, Cao Z, et al. Estrogen deficiency–mediated M2 macrophage osteoclastogenesis contributes to M1/M2 ratio alteration in ovariectomized osteoporotic mice. J Bone Miner Res. 2018;33:899–908. https://doi.org/x.1002/jbmr.3364.
-
Johnson TA, Singla DK. PTEN inhibitor VO-OHpic attenuates inflammatory M1 macrophages and cardiac remodeling in doxorubicin-induced cardiomyopathy. Am J Physiol Middle Circ Physiol. 2018;315:H1236–49. https://doi.org/x.1152/ajpheart.00121.2018.
-
Moher D, Shamseer 50, Clarke M, Ghersi D, Liberati A, Petticrew Yard, et al. Preferred reporting items for systematic review and meta-assay protocols (PRISMA-P) 2015 statement. Syst Rev. 2015;iv(1). https://doi.org/ten.1186/2046-4053-4-ane.
-
Babineau J. Product review: Covidence (systematic review software). J Can Heal Libr Assoc. 2014;35:68.
-
Wells K, Littell JH. Study quality assessment in systematic reviews of research on intervention furnishings. Res Soc Work Pract. 2009;19:52–62.
-
Borges GÁ, Rêgo DF, Assad DX, Coletta RD, De Luca Canto 1000, Guerra ENS. In vivo and in vitro furnishings of curcumin on head and cervix carcinoma: a systematic review. J Oral Pathol Med. 2017;46:iii–20. https://doi.org/x.1111/jop.12455.
-
Hong-Tao X, Chien-Wei 50, Yan LM, Yu-Fan Westward, Shu-Hang Patrick Y, Kuang-Sheng OL. The shift in macrophages polarization afterward tendon injury: a systematic review. J Orthop Transl. 2019;21:24–34. https://doi.org/10.1016/j.jot.2019.11.009.
-
Shahmohammadi K, Shariatpanahi ZV, Hajimohammadebrahim-Ketabforoush One thousand. In vitro and in vivo antitumor activity of vitamin D3 in malignant gliomas: a systematic review. Int J Cancer Manag. 2020;13(two):e94542. https://doi.org/ten.5812/ijcm.94542.
-
Mahon OR, Kelly DJ, McCarthy GM, Dunne A. Osteoarthritis-associated basic calcium phosphate crystals change immune cell metabolism and promote M1 macrophage polarization. Osteoarthr Cartil. 2019;S1063-4584(1019):31247–6. https://doi.org/10.1016/j.joca.2019.ten.010.
-
Zhou F, Mei J, Han 10, Li H, Yang S, Wang Yard, et al. Kinsenoside attenuates osteoarthritis by repolarizing macrophages through inactivating NF-kappa B/MAPK signaling and protecting chondrocytes. Acta Pharm Sin B. 2019;9:973–85. https://doi.org/10.1016/j.apsb.2019.01.015.
-
Jablonski CL, Leonard C, Salo P, Krawetz RJ. CCL2 but not CCR2 is required for spontaneous articular cartilage regeneration post-injury. J Orthop Res. 2019;37:2561–74. https://doi.org/10.1002/jor.24444.
-
Yarnall BW, Chamberlain CS, Hao Z, Muir P. Proinflammatory polarization of stifle synovial macrophages in dogs with cruciate ligament rupture. Vet Surg. 2019;48:1005–12. https://doi.org/x.1111/vsu.13261.
-
Haltmayer Eastward, Ribitsch I, Gabner S, Rosser J, Gueltekin South, Peham J, et al. Co-culture of osteochondral explants and synovial membrane as in vitro model for osteoarthritis. PLoS One. 2019;14:one–19. https://doi.org/x.1371/journal.pone.0214709.
-
Liu B, Zhang Chiliad, Zhao J, Zheng M, Yang H. Imbalance of M1/M2 macrophages is linked to severity level of knee osteoarthritis. Exp Ther Med. 2018;sixteen:5009–xiv. https://doi.org/10.3892/etm.2018.6852.
-
Sambamurthy N, Zhou C, Nguyen V, Smalley R, Hankenson KD, Dodge GR, et al. Deficiency of the blueprint-recognition receptor CD14 protects against joint pathology and functional reject in a murine model of osteoarthritis. PLoS One. 2018;e206217:13. https://doi.org/ten.1371/journal.pone.0206217.
-
Wang W, Lin 10, Xu H, Sun Due west, Bouta EM, Zuscik MJ, et al. Attenuated joint tissue impairment associated with improved synovial lymphatic function following treatment with Bortezomib in a mouse model of experimental posttraumatic osteoarthritis. Arthritis Rheumatol. 2019;71:244–57. https://doi.org/10.1002/art.40696.
-
Zhang H, Lin C, Zeng C, Wang Z, Wang H, Lu J, et al. Synovial macrophage M1 polarisation exacerbates experimental osteoarthritis partially through R-spondin-2. Ann Rheum Dis. 2018;77(10):1524–34. https://doi.org/10.1136/annrheumdis-2018-213450.
-
Timur UT, Caron MMJ, Bastiaansen-Jenniskens YM, Welting TJM, van Rhijn LW, van Osch GJVM, et al. Celecoxib-mediated reduction of prostanoid release in Hoffa's fat pad from donors with cartilage pathology results in an attenuated inflammatory phenotype. Osteoarthr Cartil. 2018;26:697–706. https://doi.org/10.1016/j.joca.2018.01.025.
-
Topoluk Northward, Steckbeck K, Siatkowski S, Burnikel B, Tokish J, Mercuri J. Amniotic mesenchymal stalk cells mitigate osteoarthritis progression in a synovial macrophage-mediated in vitro explant coculture model. J Tissue Eng Regen Med. 2018;12:1097–110. https://doi.org/ten.1002/term.2610.
-
Manferdini C, Paolella F, Gabusi Due east, Gambari L, Piacentini A, Filardo G, et al. Adipose stromal cells mediated switching of the pro-inflammatory profile of M1-like macrophages is facilitated past PGE2: in vitro evaluation. Osteoarthr Cartil. 2017;25:1161–71. https://doi.org/10.1016/j.joca.2017.01.011.
-
Pal South, Konkimalla VB. Sulforaphane regulates phenotypic and functional switching of both induced and spontaneously differentiating human monocytes. Int Immunopharmacol. 2016;35:85–98. https://doi.org/10.1016/j.intimp.2016.03.008.
-
Siebelt M, Korthagen Northward, Wei W, Groen H, Bastiaansen-Jenniskens Y, Muller C, et al. Triamcinolone acetonide activates an anti-inflammatory and folate receptor-positive macrophage that prevents osteophytosis in vivo. Arthritis Res Ther. 2015;17:352. https://doi.org/x.1186/s13075-015-0865-1.
-
Fahy Northward, de Vries-van Melle ML, Lehmann J, Wei W, Grotenhuis N, Farrell E, et al. Human osteoarthritic synovium impacts chondrogenic differentiation of mesenchymal stem cells via macrophage polarisation state. Osteoarthr Cartil. 2014;22:1167–75. https://doi.org/10.1016/j.joca.2014.05.021.
-
Tsuneyoshi Y, Tanaka M, Nagai T, Sunahara N, Matsuda T, Sonoda T, et al. Functional folate receptor beta-expressing macrophages in osteoarthritis synovium and their M1/M2 expression profiles. Scand J Rheumatol. 2012;41:132–40. https://doi.org/ten.3109/03009742.2011.605391.
-
Zhang S, Chuah SJ, Lai RC, Hui JHP, Lim SK, Toh WS. MSC exosomes mediate cartilage repair by enhancing proliferation, attenuating apoptosis and modulating immune reactivity. Biomaterials. 2018;156:sixteen–27. https://doi.org/x.1016/j.biomaterials.2017.11.028.
-
Hu Y, Gui Z, Zhou Y, Xia L, Lin K, Xu Y. Quercetin alleviates rat osteoarthritis by inhibiting inflammation and apoptosis of chondrocytes, modulating synovial macrophages polarization to M2 macrophages. Free Radic Biol Med. 2019;145:146–60. https://doi.org/10.1016/j.freeradbiomed.2019.09.024.
-
Dai Chiliad, Sui B, Xue Y, Liu X, Sun J. Cartilage repair in degenerative osteoarthritis mediated by squid type Two collagen via immunomodulating activation of M2 macrophages, inhibiting apoptosis and hypertrophy of chondrocytes. Biomaterials. 2018;180:91–103. https://doi.org/10.1016/j.biomaterials.2018.07.011.
-
Barreto G, Senturk B, Colombo L, Brück O, Neidenbach P, Salzmann Chiliad, et al. Lumican is upregulated in osteoarthritis and contributes to TLR4-induced pro-inflammatory activation of cartilage degradation and macrophage polarization. Osteoarthr Cartil. 2020;28:92–101. https://doi.org/x.1016/j.joca.2019.10.011.
-
Kraus VB, McDaniel Chiliad, Huebner JL, Stabler Television, Pieper CF, Shipes SW, et al. Direct in vivo evidence of activated macrophages in human osteoarthritis. Osteoarthr Cartil. 2016;24:1613–21. https://doi.org/x.1016/j.joca.2016.04.010.
-
Utomo L, van Osch GJVM, Bayon Y, Verhaar JAN, Bastiaansen-Jenniskens YM. Guiding synovial inflammation by macrophage phenotype modulation: an in vitro report towards a therapy for osteoarthritis. Osteoarthr Cartil. 2016;24:1629–38. https://doi.org/10.1016/j.joca.2016.04.013.
-
Alvarado-Vazquez PA, Bernal L, Paige CA, Grosick RL, Moracho Vilrriales C, Ferreira DW, et al. Macrophage-specific nanotechnology-driven CD163 overexpression in human macrophages results in an M2 phenotype nether inflammatory atmospheric condition. Immunobiology. 2017;222:900–12. https://doi.org/10.1016/j.imbio.2017.05.011.
-
Miyaji Northward, Nishida One thousand, Tanaka T, Araki D, Kanzaki North, Hoshino Y, et al. Inhibition of knee osteoarthritis progression in mice past administering SRT2014, an activator of silent information regulator 2 Ortholog 1. Cartilage. 2020:1947603519900795. https://doi.org/10.1177/1947603519900795.
-
Menarim BC, Gillis KH, Oliver A, Bricklayer C, Werre SR, Luo 10, et al. Inflamed synovial fluid induces a homeostatic response in bone marrow mononuclear cells in vitro: implications for joint therapy. FASEB J. 2020;34:4430–44. https://doi.org/10.1096/fj.201902698R.
-
Zhou F, Mei J, Yang S, Han X, Li H, Yu Z, et al. Modified ZIF-8 nanoparticles attenuate osteoarthritis past reprogramming the metabolic pathway of synovial macrophages. ACS Appl Mater Interfaces. 2020;12(ii):2009–22. https://doi.org/10.1021/acsami.9b16327.
-
Shu CC, Zaki S, Ravi 5, Schiavinato A, Smith MM, Little CB. The relationship betwixt synovial inflammation, structural pathology, and pain in mail-traumatic osteoarthritis: differential issue of stem cell and hyaluronan treatment. Arthritis Res Ther. 2020;22:1–xiii. https://doi.org/10.1186/s13075-015-0865-i.
-
Nicholson LB. The allowed organization. Essays Biochem. 2016;60:275–301.
-
Watanabe S, Alexander M, Misharin AV, Budinger GRS. The role of macrophages in the resolution of inflammation. J Clin Invest. 2019;129:2619–28. https://doi.org/ten.1172/JCI124615.
-
Wenham CYJ, Conaghan PG. The role of synovitis in osteoarthritis. Ther Adv Musculoskelet Dis. 2010;2:349–59. https://doi.org/10.1177/1759720X10378373.
-
Griffin TM, Scanzello CR, Griffin TM, Griffin TM. Innate inflammation and synovial macrophages in osteoarthritis pathophysiology. Clin Exp Rheumatol. 2019;37 Suppl 120(5):57–63.
-
Clockaerts S, Bastiaansen-Jenniskens YM, Runhaar J, Van Osch GJVM, Van Offel JF, Verhaar JAN, et al. The infrapatellar fatty pad should exist considered every bit an agile osteoarthritic articulation tissue: a narrative review. Osteoarthr Cartil. 2010;eighteen:876–82. https://doi.org/10.1016/j.joca.2010.03.014.
-
Glasson SS, Blanchet TJ, Morris EA. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthr Cartil. 2007;15:1061–9. https://doi.org/10.1016/j.joca.2007.03.006.
-
Magri C, Schramme Thousand, Febre M, Cauvin E, Labadie F, Saulnier N, et al. Comparison of efficacy and safety of unmarried versus repeated intra-articular injection of allogeneic neonatal mesenchymal stem cells for handling of osteoarthritis of the metacarpophalangeal/metatarsophalangeal joint in horses: a clinical airplane pilot written report. Fiorina P, editor PLoS One 2019;xiv:e0221317. doi:https://doi.org/ten.1371/journal.pone.0221317.
-
Wynn TA, Barron Fifty, Thompson RW, Madala SK, Wilson MS, Cheever AW, et al. Quantitative assessment of macrophage functions in repair and fibrosis. Curr Protoc Immunol. 2011;Chapter 14:fourteen.22. https://doi.org/10.1002/0471142735.im1422s93.
-
Croft AP, Campos J, Jansen Yard, Turner JD, Marshall J, Attar Yard, et al. Distinct fibroblast subsets bulldoze inflammation and damage in arthritis. Nature. 2019;570:246–51. https://doi.org/10.1038/s41586-019-1263-vii.
-
Culemann S, Grüneboom A, Nicolás-Ávila JÁ, Weidner D, Lämmle KF, Rothe T, et al. Locally renewing resident synovial macrophages provide a protective barrier for the joint. Nature. 2019;572(7771):670–five. https://doi.org/x.1038/s41586-019-1471-1.
-
Cosenza South, Ruiz M, Toupet K, Jorgensen C, Noël D. Mesenchymal stem cells derived exosomes and microparticles protect cartilage and bone from deposition in osteoarthritis. Sci Rep. 2017;7:1–12. https://doi.org/ten.1038/s41598-017-15376-8.
-
Kong Fifty, Zheng LZ, Qin L, Ho KKW. Part of mesenchymal stalk cells in osteoarthritis treatment. J Orthop Transl. 2017;ix:89–103. https://doi.org/10.1016/j.jot.2017.03.006.
-
Chahal J, Gómez-Aristizábal A, Shestopaloff Grand, Bhatt Southward, Chaboureau A, Fazio A, et al. Bone marrow mesenchymal stromal cells in patients with osteoarthritis results in overall improvement in pain and symptoms and reduces synovial inflammation. Stem Cells Transl Med. 2019;8(8):746–57. https://doi.org/10.1002/sctm.18-0183.
-
Hofer HR, Tuan RS. Secreted trophic factors of mesenchymal stalk cells support neurovascular and musculoskeletal therapies. Stalk Prison cell Res Ther. 2016;seven:1–14. https://doi.org/x.1186/s13287-016-0394-0.
-
Yeadon C, Karsh J. Lymphapheresis in rheumatoid arthritis. The clinical and laboratory furnishings of a limited class of cell depletion. Clin Exp Rheumatol. 1983;1:119–24.
-
Hahn G, Stuhlmüller B, Hain N, Kalden JR, Pfizenmaier Thou, Burmester GR. Modulation of monocyte activation in patients with rheumatoid arthritis past leukapheresis therapy. J Clin Invest. 1993;91:862–70. https://doi.org/10.1172/JCI116307.
-
Degboé Y, Rauwel B, Businesswoman K, Boyer J-F, Ruyssen-Witrand A, Constantin A, et al. Polarization of rheumatoid macrophages past TNF targeting through an IL-10/STAT3 mechanism. Front Immunol. 2019;10:3. https://doi.org/10.3389/fimmu.2019.00003.
-
Tardito S, Martinelli G, Soldano S, Paolino S, Pacini G, Patane Thousand, et al. Macrophage M1/M2 polarization and rheumatoid arthritis: a systematic review. Autoimmun Rev. 2019;18:102397. https://doi.org/x.1016/j.autrev.2019.102397.
-
Blom AB, van Lent PLEM, Holthuysen AEM, van der Kraan PM, Roth J, van Rooijen N, et al. Synovial lining macrophages mediate osteophyte germination during experimental osteoarthritis. Osteoarthr Cartil. 2004;12:627–35. https://doi.org/10.1016/j.joca.2004.03.003.
-
He Due west, Kapate N, Shields CW, Mitragotri S. Drug delivery to macrophages: a review of targeting drugs and drug carriers to macrophages for inflammatory diseases. Adv Drug Deliv Rev. 2020;165-166:15–twoscore. https://doi.org/x.1016/j.addr.2019.12.001.
-
Wang LX, Zhang SX, Wu HJ, Rong Xl, Guo J. M2b macrophage polarization and its roles in diseases. J Leukoc Biol. 2019;106:345–58. https://doi.org/10.1002/JLB.3RU1018-378RR.
-
MacParland SA, Tsoi KM, Ouyang B, Ma Ten-Z, Manuel J, Fawaz A, et al. Phenotype determines nanoparticle uptake past human macrophages from liver and blood. ACS Nano. 2017;11:2428–43. https://doi.org/10.1021/acsnano.6b06245.
-
Bianchini R, Roth-Walter F, Ohradanova-Repic A, Flicker S, Hufnagl K, Fischer MB, et al. IgG4 drives M2a macrophages to a regulatory M2b-like phenotype: potential implication in immune tolerance. Allergy. 2019;74:483–94. https://doi.org/10.1111/all.13635.
-
Liberale L, Dallegri F, Montecucco F, Carbone F. Pathophysiological relevance of macrophage subsets in atherogenesis. Thromb Haemost. 2017;117:07–eighteen. https://doi.org/10.1160/TH16-08-0593.
-
Wynn TA, Chawla A, Pollard JW. Macrophage biology in development, homeostasis and disease. Nature. 2013;496:445–55. https://doi.org/10.1038/nature12034.
-
Xue J, Schmidt SV, Sander J, Draffehn A, Krebs W, Quester I, et al. Transcriptome-based network assay reveals a spectrum model of human macrophage activation. Immunity. 2014;twoscore:274–88. https://doi.org/10.1016/j.immuni.2014.01.006.
-
Avraham R, Haseley N, Brown D, Penaranda C, Jijon HB, Trombetta JJ, et al. Pathogen cell-to-cell variability drives heterogeneity in host immune responses. Cell. 2015;162:1309–21. https://doi.org/10.1016/j.cell.2015.08.027.
-
Lavin Y, Winter D, Blecher-Gonen R, David E, Keren-Shaul H, Merad Chiliad, et al. Tissue-resident macrophage enhancer landscapes are shaped past the local microenvironment. Cell. 2014;159:1312–26. https://doi.org/10.1016/j.cell.2014.11.018.
-
Misharin AV, Morales-Nebreda L, Reyfman PA, Cuda CM, Walter JM, McQuattie-Pimentel AC, et al. Monocyte-derived alveolar macrophages drive lung fibrosis and persist in the lung over the life span. J Exp Med. 2017;214:2387–404. https://doi.org/ten.1084/jem.20162152.
-
Reyfman PA, Walter JM, Joshi Northward, Anekalla KR, McQuattie-Pimentel AC, Chiu Due south, et al. Single-cell transcriptomic analysis of man lung provides insights into the pathobiology of pulmonary fibrosis. Am J Respir Crit Care Med. 2019;199:1517–36. https://doi.org/10.1164/rccm.201712-2410OC.
-
Fang H, Beier F. Mouse models of osteoarthritis: modelling take a chance factors and assessing outcomes. Nat Rev Rheumatol. 2014;10:413–21. https://doi.org/ten.1038/nrrheum.2014.46.
Acknowledgements
This written report was supported by the fundings of YZJ (the National Cardinal R&D Program of China, 2019YFA0111900 to YJ), OKL (MWLC Associate Member Program, Ming Wai Lau Eye of Reparative Medicine of Karolinska Institute to LOK), and CWL (Hong Kong Regime Enquiry Grant Council, General Research Fund Reference no. 14104620 to CWL).
Funding
This study was supported past the Ministry of Scientific discipline and Applied science (the National Central R&D Program of China, 2019YFA0111900 to YJ), MWLC Associate Member Plan, Ming Wai Lau Centre of Reparative Medicine of Karolinska Constitute to LOK., and also by Hong Kong Government Research Grant Council, Full general Research Fund (Reference no. 14104620 to CWL).
Author data
Affiliations
Contributions
All authors listed accept read and approved all versions of the manuscript. Conception and design: OKL, XBZ, and HTX. Drafting of the article: XBZ and YFW. Critical revision of the article for of import intellectual content: XBZ, OKL, PSY, YZJ, and CWL. The authors read and canonical the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Non applicable.
Competing interests
The authors declare that they have no competing interests.
Boosted information
Publisher'south Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Boosted file i: Supplemental 1a.
Methodological quality assessment protocol i used for included animal studies (the number of "yes" answers was counted for each written report to give a full score out of 8). Supplemental 1b. Quality assessment for systemic reviews in experimental brute studies. Supplemental 2. Risk of Bias Assessment for in vitro studies according to GRADE Criteria
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits apply, sharing, adaptation, distribution and reproduction in whatever medium or format, as long equally you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other 3rd political party material in this article are included in the article'south Creative Eatables licence, unless indicated otherwise in a credit line to the textile. If material is not included in the commodity's Artistic Commons licence and your intended employ is non permitted by statutory regulation or exceeds the permitted employ, you will need to obtain permission directly from the copyright holder. To view a re-create of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and Permissions
About this article
Cite this commodity
Zhu, 10., Lee, CW., Xu, H. et al. Phenotypic alteration of macrophages during osteoarthritis: a systematic review. Arthritis Res Ther 23, 110 (2021). https://doi.org/10.1186/s13075-021-02457-3
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s13075-021-02457-3
Keywords
- Osteoarthritis
- Macrophage
- Innate amnesty
- Immunomodulation
Source: https://arthritis-research.biomedcentral.com/articles/10.1186/s13075-021-02457-3
0 Response to "what phenotypic changes occur to the macrophages that cause them to be anti-inflammatory"
Post a Comment